In the wake of The Garden Professors’ sudden notoriety (see Linda’s Jan. 26 post), my department head sent out a very kind e-mail announcement to our faculty, staff, and grad students.
However, he referred to us as the "Hort Professors" blog, sans hyperlink.
A curious staff member (the lovely and talented Pris Sears) searched that title, resulting in the following:
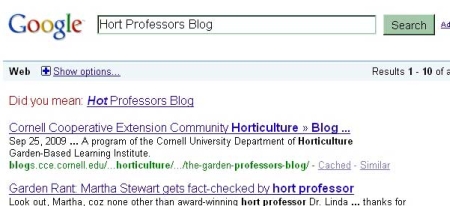
Hort Professors, Hot Professors…kind of the same thing. Thanks, Google!

