One of the newer “miracle products” targeted to gardeners is rock dust. Rock dust (also called rock flour or rock mineral powder) is exactly what it sounds like. It is a byproduct of quarry work and is generally a finely pulverized material that resembles silt. It’s heavily promoted as a way to provide macro- and micronutrients to your soils and plants. Is it worth adding to your gardens?

First, it’s worth acknowledging that repurposing an industry byproduct is always preferable to throwing it away. Fortunately, the last few years have yielded some peer-reviewed research that we can use to make informed recommendations.
What’s in rock dust?
Obviously, the mineral content of rock dust is dependent on the rocks used to make it. This means the mineral content varies considerably, but in general rock dusts contain:
- Large amounts of silicon, aluminum, and sometimes iron
- Lesser amounts of calcium, copper, magnesium, manganese, potassium, sulfur, and zinc.
- Potentially toxic levels of aluminum, arsenic, cadmium, chromium, copper, lead, nickel, and sodium.
I’ve added some tables from a few research articles that analyzed their rock dust mineral content below. Note the high silcon, aluminum, and iron content. (LOI = loss on ignition, meaning some materials were burned off during analysis.)



How is rock dust used as a mineral source?
Rock dusts must be solubilized to release minerals. There are some criteria that can speed mineral release:
- Decreasing the particle size of rock dust.
- Blending the rock dust with nutrient-rich organic matter like manure. This provides an acidified environment for mineral solubilization.
When is it beneficial to use rock dust?
There are documented benefits to using rock dusts – but only in agricultural production systems:
- Rock dusts can contribute minerals to nutrient depleted soils, such as agricultural soils that have been overworked for decades.
- Organic farmers can use specific rock dusts to supply micronutrients, rather than commercial fertilizers which are not certified for organic crop production.
- Cereal crops – members of the grass family – require silica as a micronutrient (though silica is rarely if ever deficient in field conditions).
What’s the bottom line for gardeners?
As one article states, “…there is a potential for using [rock flour]…where there is a lack of these nutrients and where conventional chemical fertilizers are either not available or not desired.”
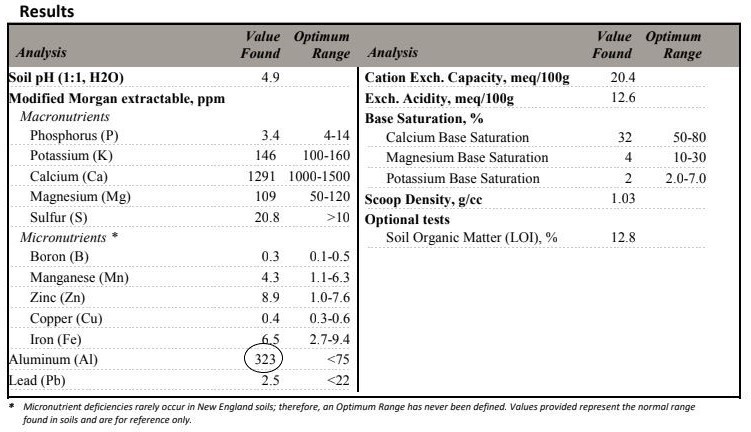
And how do you know if you have a lack of a certain nutrient? Why, by having your soil tested, of course! There is no point in adding anything to your soil unless something is missing. It is MUCH harder to treat a nutrient toxicity than to add a deficient nutrient. Iif a soil test reveals a lack of a particular nutrient, a carefully chosen product could supply this mineral. But you would have to know what else was being supplied and possibly creating a mineral toxicity.
At this point, there is no evidence to suggest that rock dusts are of any value to a home garden or landscape. And adding these products can easily contribute to aluminum and heavy metal toxicities. I would never add it to this soil, for instance, as it already has excessively high aluminum levels.
This blog is full of great ideas on how to manage your soil naturally, sustainably, and safely. Rock dusts are just the latest garden product with lots of marketing but little benefit.



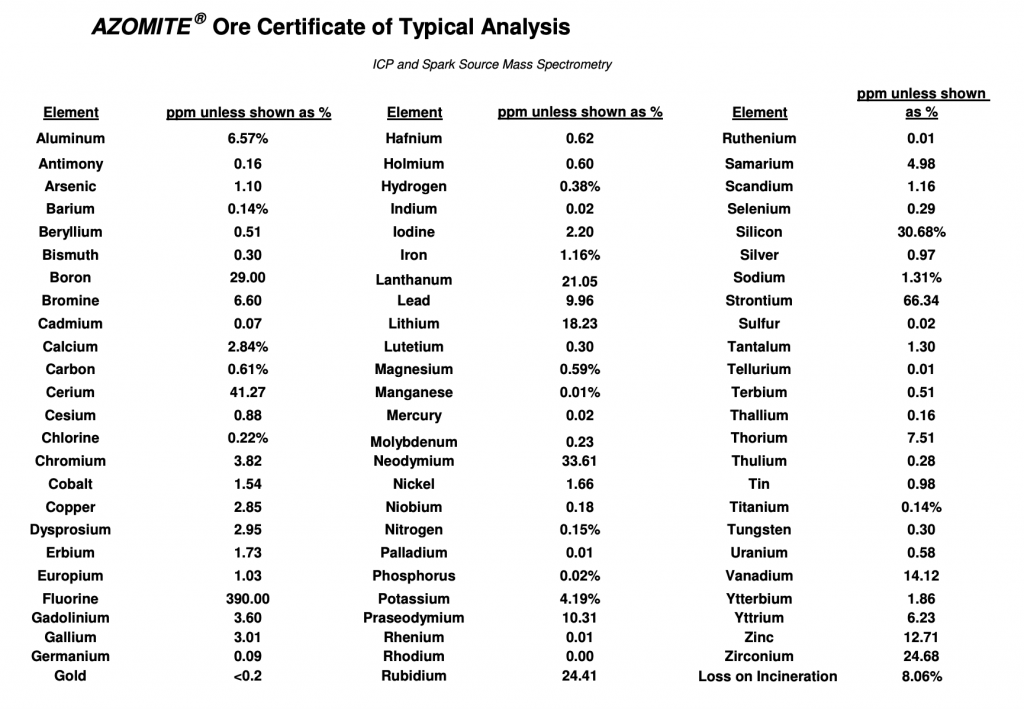
“Sounds like we need to update our plant nutrition textbooks.” I learned the top 3 + 11 more as essential for plant nutrition – what are the others? It occurs to me that this might be sarcasm, since I’ve read other critiques of the dubious Azomite (fav of the weed growers). But maybe you could make that clear otherwise definitely you’re going to be (mis)quoted!
Just found this: https://www.compostwerks.com/images/Azomite_Analysis.pdf so now I understand your comment.
How can Oxygen or Hydrogen be a trace element in rock powder? aren’t those gases? (I never took chemistry!) BUT I do like that the elements have been found “with sophisticated scientific analytical methods.”
Hi Kathie –
Yes, it’s sarcasm. Most of the minerals are not required by any plant for any process, and several are toxic heavy metals that will compromise enzymatic systems in all forms of life.
Currently there are 17 known essential nutrients for all plants, and 4 others that are required by certain plants (they are in parentheses below). I like to group them by functionality:
C, H and O – basic structural building blocks
N, P and S – protein, nucleic acid, and membrane structual building blocks
B, Ca and (Si) – cell walls and membranes structure and function
Cl, K and (Na) – water management
(Co) Cu, Fe, Mg, Mn, Mo, Ni, (Se) and Zn – enzyme co-factors
Hi Kathie,
I’ve just found this blog, and saw your comment. I can at least answer your question about oxygen and hydrogen. You are correct that in their native state that they are gasses, but once they are bound into molecules, they may no longer be gaseous.
For instance, think about table salt – it’s made out of sodium chloride. If you used these ‘sophisticated scientific analytical methods’ you would find that table salt is made up of about 60% chlorine (by weight). But chlorine is a gas, yet table salt is a solid. It’s the bonding between the sodium and the chlorine that effects this property.
Same goes with the hydrogen (or oxygen, nitrogen etc) in rock powder – it’s present in the bonded form. Many minerals contain these elements – oxygen is very very common in rocks (if you look closely at the table, you’ll see the major elements are listed as oxides – SiO2, Al2O3 etc – the O in those formulas is oxygen). Hydrogen is also relatively common – clay minerals all contain hydrogen, as do many other common minerals (e.g. micas, amphiboles and a lot of others).
The table tells you the chemical make up of the rocks, but it doesn’t really tell you how those elements are bonded together. That will depend on the minerals that are present.
Hope that answers your question (or for anybody else who stumbles on this comment).
thanks!
Hello Linda, Thank you so much for addressing the question of rock dusts! All too often it is the fact that someone has something they want to sell that grabs public attention, not whether anyone needs to spend money on it. When I was teaching horticulture, I always cautioned students to be suspicious of a product unless they could find a recommendation for it from sources that weren’t selling it. (This is true in so many fields. I see many “new” food products marketed as the elixir of health simply because someone wants to sell them.) I had reached similar conclusions to the ones you reached about rock dust, but it is extremely useful to the public to have your analysis.
I have a question for you in reference to mulches. I do a lot of teaching for the Master Gardener programs on creating fire-resistant landscape. I have read and wrote many article on fire resistant vegetation. I agree with some of the earlier post about the concerns with lists of firewise/firesafe plants. I try to inform people on the attributes of plants that are less likely to burn, or produce less energy in a burning environment. An area that I have tried to find information on is scoria/cinders. I have not been able to find any research on scoria concerning it value, if any to the soil. Here are some attributes of scoria that I have shared with people. it does not burn, it holds in moisture, reduces heat to the soil, adds some nutrients to the soil over time, does not have to be replaced annually, it doesn’t blow away like woody products. I see this as a great alternative to wood products close to a structure. I have not seen any research on this geologic resource. Can you direct me to a source or do you have any information on scoria and its value in a fire-resistant landscape.
The best thing to do is search on Google scholar. If you don’t find anything there it’s doubtful any research exists on the use as you describe.
Hi Linda,
Just found your blog. Really enjoying a scientific analysis of gardening. As a scientist and gardener, I’m frustrated by the amount of nonsense that’s perpetuated from one person to the next.
Just looking at this though, I think that what would mater more than the total Al content in the additive would be the form of that Al. Al2O3 is incredibly insoluble – it’s about as insoluble as something can get under surface conditions. Al is the most abundant metal in the continental crust (~8%), which as you’ve correctly said would be toxic. But obviously this hasn’t prevented plants living on the continental crust – it’s not bioavailable because it’s too immobile.
A chemical analysis like this doesn’t ever tell the full story. It’s not just the presence of the elements, but their form as well.
Interesting that you mentioned Si too – I had a colleague who did some trials with adding Si to soils – it was actually dunite (a typical low silica igneous rock, still ~40% SiO2) that added the most silica to the soil, not the high silica rocks (like diatomite, ~99% SiO2). The mineralogy played a bigger effect than the chemistry. I don’t think it was ever published, it was part of a commercial operation he was running.
Soil testing involves chemical extractions that are not necessarily relevant to the plants and microbes in the system. Roots and fungi are well-adapted to solubilizing minerals for uptake – after all, that’s the way that solid rock is eventually turned to soil.
You really need to consider a study on Volcanic Rock Dust which I first began using around 30 years so it’s not all that new in Michigan. It makes a truly remarkable difference in my garden.
There’s no research that shows that rock dust of any sort makes any difference. Without a structured, replicated experiment comparing part of your garden to another part (one treated, one not) you don’t really know what’s causing the difference. There are too many natural variables that can affect garden productivity from year to year.
“New to me is that plants require 67 essential nutrients?” Have you never heard of Liebig’s Law of the Minimum? This Law states that the local yield of a plants is limited by the nutrient that is present in the environment in the “least” quantity relative to its demands for plant growth. This statement has been confirmed worldwide. Justis von Liebig was considered the “father of the fertilizer industry” for his emphasis on nitrogen and trace minerals as essential plant nutrients.
Your response to Norma Jean is ill-informed. There are multiple studies by scientists and agronomists from all over the world on volcanic ash’s positive impact on soil health, increased brix, improved yields.
Lastly, Azomite is not a ‘repurposed industry byproduct’ — it is volcanic ash and is not considered a rock dust in the first place.
I’m also a doctor with my PhD in woody plant physiology. I’m well versed in what nutrients are considered essential and why. Since you are a medical doctor, I expect you know what the essential nutrients for animals are. Somehow I doubt that list has expanded to 67 as well. Just because you can find a variety of elements in a plant (or a human) does not make them “essential.”
As a doctor and, I presume, a scientist, I’m sure you understand the necessity to provide peer-reviewed evidence to support your points. Simply saying “multiple studies by scientists and agronomists” doesn’t cut it. Feel free to provide literature citations if you have them.