California had the worst fires in the last two years of its existence as a state. Hundreds of thousands of acres of brush and forest burned. More importantly thousands lost their homes as fires moved across urban/rural interfaces to destroy communities. The entire town of Paradise, California was burned to the ground. Here in Ventura County, the Thomas Fire was the state’s largest fire by the time it was done, and hundreds lost homes. No other time in history have we been so focused on what will burn, why it will burn, and what we can do to have a “firewise” landscape.

Fire authorities around the world have advocated creating defensible spaces around homes that are clear of ignitable vegetation. Some authorities have mandated by law that mulch, pine needles and other debris be removed as a fire prevention measure near structures. There is a general recognition that any plant can burn. Even well irrigated plants will rapidly desiccate and become flammable in the face of strong wind and a heavy fuel load that is inflamed nearby.
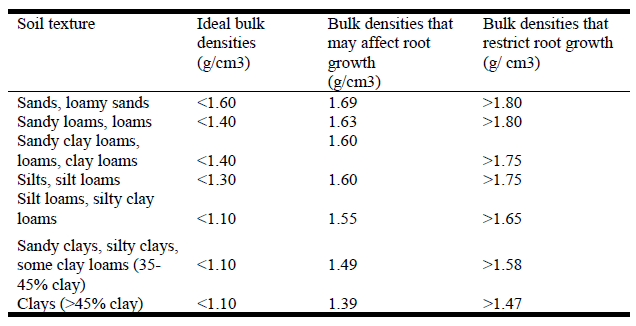
Flammability of landscape around homes is dependent on several factors. Vegetation placement can obstruct or allow for fire fighters ability to reduce damage to a home. While it is natural to assume that avoiding flammable plants is a part of this process, there is no standard method for testing plant flammability. Many lists of firewise plants have unknown origin or are just guesses. Flammability can be assigned four dimensions: ignitability, sustainability, combustibility and consumability. These factors refer to time till ignition; time a material will burn; rapidity or intensity of burn and quantity of material that will burn. The components of combustion are influenced by moisture content, percentage of carbon, percentage of volatile compounds; surface area to volume ratio and other factors. The varied factors are usually not all studied at the same time and are not all equally important to plant flammability. Thus assessing flammability even within the context of a controlled study will only partially assess a material’s likelihood of burning under various conditions. Hence most of the lists are not that helpful.

Behm et. al. showed that variations in flammability between plant species exists, and also that species within the same genus can vary widely in their flammable nature. Thus lists should not assume species in the same genus all have the same flammability. There is some thinking that flammability is an evolutionary trait that some plants exploit to their benefit, i.e. they are made to burn, such as the California Chaparral plant communities. Simply burning fuels in a laboratory setting does not take into account many of the factors associated with fuel burning intensities. Species differences notwithstanding, the amount of dead plant matter (dead twigs and leaves) vs. live matter, the arrangement of leaves, mulch and adjacent species all play a role in the flammability of the landscape itself which cannot be studied in a lab setting. Landscapes are “fuel bed complexes” with multiple elements that are not replicated in studies. For instance, small leaves from some shrubs ignite easily, but when burned as litter, develop low heat release rates because of poor ventilation.
Testing live plant materials alone is misleading because the flammability of an intact shrub is caused by the interaction of live matter with “necromatter”. Dead tissues are thermal catalysts which ignite live material. The ratio of necromatter to live matter influences flammability and is generally not well studied. Fire modelling also has a role in understanding what will burn. Both wind and slope increase the spread rate and the fireline intensity of burnable plants. Fire behavior characteristics on a given plant also are affected by both its physical and chemical characteristic — tissue mineral and water content have impacts on flammability. This bodes poorly for firesafe plant lists because lists do not consider plant physical or chemical attributes and if moisture levels are low it will burn regardless of its structure and geometry or its status on a list. Sometimes though a dense wall of well hydrated vegetation can save homes such as the avocado orchards that held back fire in the Thomas Fire in Montecito, Ca.
While lists don’t satisfy scientific rigor they are great for policy makers and homeowners who want to know what to plant. Unfortunately many lists are just compilations of other lists, none of which were based on research. Sometimes lists confuse one desirable characteristic with another, such as native plant lists that tout drought tolerance. Many drought tolerant plants are not fire resistant especially after a long dry period, indeed they often evolved to burn under such conditions.
For those that live in fire prone areas, fire resistant plant lists will always be an attractive or even required element of landscapes. Lists will not save a structure in the face of high winds and adequate fuel or embers. A defined defensible space around buildings, and maintenance of plantings that removes dead matter, maintains irrigation, and maintains proper distance from combustible surfaces will be more effective than choosing landscape plants from flammability lists.
References:
Fernandes, P.M. and M. G. Cruz. 2012. Plant flammability experiments offer limited insight into vegetation—fire dynamic interactions. New Phytologist 194: 606-609
Behm, A.L., M. L. Duryea, A.J. Long, and W.C. Zipperer. 2004. Flammability of native understory species in pine flatwood and hardwood hammock ecosystems and implications for the wildland –urban interface. International J. of Wildland fire 13: 355-365.
White, R.H. and W.C. Zipperer. 2010. Testing and classification of individual plants for fire behavior: plant selection for the wildland-urban interface. International J. of Wildland Fire 19:213-227.